|
The P-gp transporter is expressed on the luminal surface of the small intestine and blood-brain barrier, as well as the apical membrane of hepatocytes and kidney proximal tubule epithelia ( Fig. Inhibitors listed in the table can inhibit multiple transporters. Representative clinical DDIs are summarized in Table 1.Įlacridar is used as an oral bio-enhancer but is not an approved drug. As mentioned above, the recent FDA DDI guidance published in 2017 recommends the investigation of eight transporters for drug interactions: P-gp, BCRP, MATE1, MATE2K, OATP1B1, OATP1B3, OAT1, OAT3, and OCT2. Since then, transporter-mediated drug-drug interaction studies have increased and more transporters have been shown to have an impact on PK/PD relationships. P-gp was the first transporter included in the 2006 FDA DDI guidance documents. The importance of understanding transporter-based drug interactions has been discussed by regulatory agencies and introduced in recent FDA DDI guidance documents. Altering transporter expression and activity via drugs, dietary supplements, or genetic factors can affect the PK, PD, and organ exposure of substrates. Numerous clinical studies have demonstrated that drug transporters play important roles in altering pharmacokinetics (PK), pharmacodynamics (PD), and organ toxicity of drugs ( Fig. However, head-to-head clinical studies to validate this hypothesis have not been performed. 4–8 In summary, inhibition of FXa may represent an attractive approach compared with thrombin inhibition for effective and safe antithrombotic therapy. Early work from several laboratories provided experimental evidence from animal studies suggesting that the antithrombotic efficacy of FXa inhibitors is accompanied by a lower risk of bleeding when compared with thrombin inhibitors. These small amounts of thrombin might be enough to activate high-affinity platelet thrombin receptors to preserve hemostasis. In addition, reversible FXa inhibitors might not completely suppress the production of thrombin. 3 Second, inhibition of FXa is not thought to affect existing levels of thrombin and its activity. 
This principle is consistent with an in vitro observation that inhibition of FXa produced a more effective sustained reduction of thrombus-associated procoagulant activity than inhibition of thrombin activity. 2 In theory, therefore, inhibition of FXa may be more efficient in reducing fibrin formation than direct inhibition of thrombin activity. 
However, there is some theoretical and preclinical evidence to support that the FXa mechanism may positively differentiate from thrombin as a preferred antithrombotic target.įirst, as blood coagulation involves sequential steps of activation and amplification of coagulation proteins, generation of one molecule of FXa results in the production of hundreds of thrombin molecules.  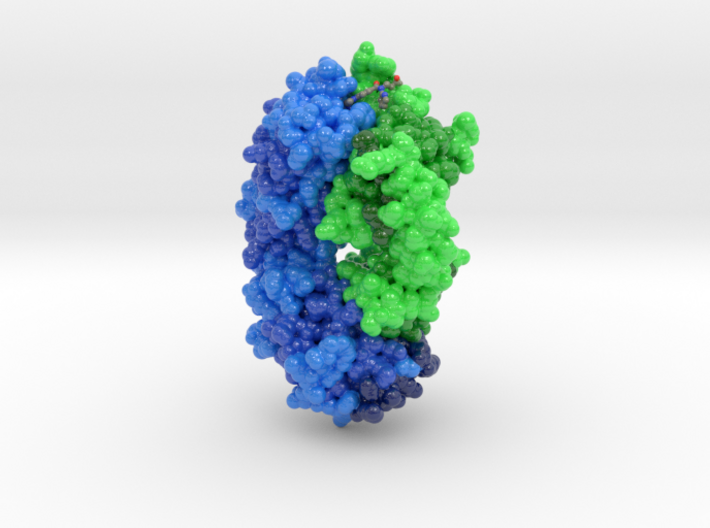
1–4 To date, there is no direct clinical evidence favoring one target over the other. Direct thrombin inhibitors, such as dabigatran etexilate, and FXa inhibitors, such as rivaroxaban and apixaban, have been developed and shown to be effective oral anticoagulants. Vitamin K antagonists (VKAs), such as warfarin, are no longer the only available oral anticoagulants. Wexler, in Annual Reports in Medicinal Chemistry, 2012 2 Rationale for Targeting FXa
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
